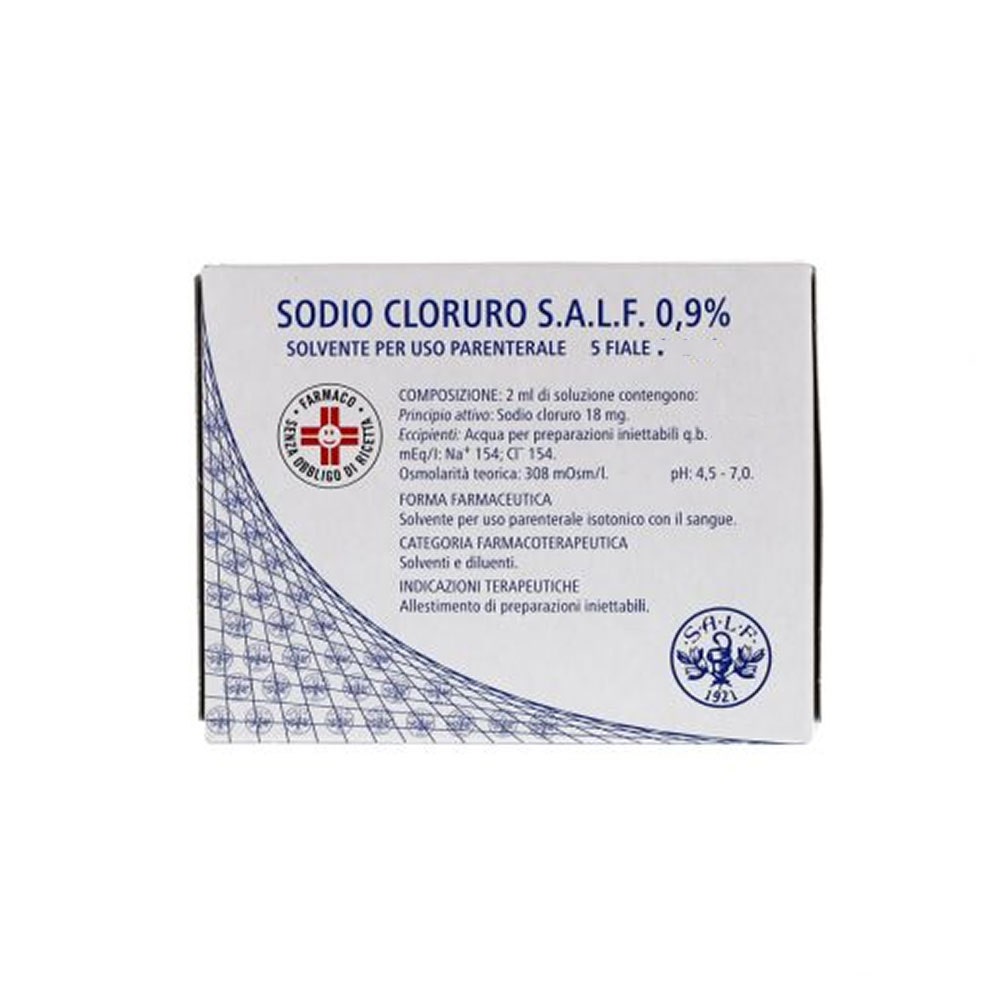
Sodium chloride 0.9% solvent for parenteral use 5 vials of 5 ml
Information on returns and shipments
Payment methods
Medicines authorized by the Ministry of Health

Sodium Chloride S.A.L.F. 0.9% is a solvent for parenteral use used for the preparation of injectable preparations.
Indications
Preparation of injectable preparations.
Composition
Active ingredients
100 ml of solution contain:
- Active ingredient: sodium chloride 0.9 g
- meq/ml: Na+ 0.154
- meq/ml: Cl- 0.154
- Osmolarity Theoretical volume (mosm/l): 308
- pH: 4.5 - 7.0
For the full list of excipients, see section 6.1.
Excipients
Water for injections.
Directions for use and Dosage
Use for extemporaneous solutions, dilutions, or suspensions of injectable medicinal products or other sterile preparations. Choose the volume to be used based on the dilution needs of the injectable preparation.
Warnings
Sodium salts should be administered with caution in patients with hypertension, cardiac failure, peripheral or pulmonary edema, impaired renal function, or impaired renal function. impaired renal function, pre-eclampsia, or other conditions associated with sodium retention (see section 4.5). The solution must be clear, colorless, and free from visible particles. Use immediately after opening the container and ensure that preparation is carried out as quickly as possible under conditions and according to procedures that preserve the sterility of the solution as much as possible. Any unused medicinal product should be discarded.
Contraindications
Hypernatremia.
Hydrosaline plethora.
For contraindications, please also refer to the SPC for the medicinal product you intend to administer.
Undesirable effects
Undesirable effects following the administration of sodium chloride are mainly related to disorders of water and electrolyte balance (hypernatremia, hyperchloremia, hypervolemia) and problems related to the administration site (infection at the infusion site, pain or local reaction, venous irritation, thrombosis or phlebitis venous thromboembolism (venous thromboembolism) extending from the infusion site, extravasation). For adverse reactions, please also refer to the SPC for the drug you intend to administer.
Overdose
The administration of excessive doses of sodium chloride solutions may lead, depending on the patient's clinical condition, to hypernatremia, hyperchloremia, and/or hypervolemia. For overdose, please also refer to the SPC for the drug you intend to administer.
Pregnancy and breastfeeding
Although no effects on fetal development have been observed, the drug should be administered only if strictly necessary and only after evaluating the risk/benefit ratio. The drug is compatible with breastfeeding. For precautions to be taken during pregnancy and breastfeeding, please also refer to the product information for the medicinal product you intend to administer.
Storage
Store in a tightly closed container. Do not freeze or refrigerate. For storage conditions after dilution with the medicinal product to be administered, please refer to section 6.4 of the Summary of Product Characteristics of the medicinal product you intend to administer.
Format
5 Vials of 5 ml
Price Trend
This product has been on sale since 08/01/2026
In the last 30 days, the product's lowest price was 4,14 €