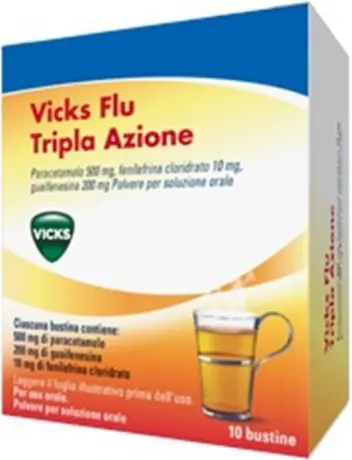
Flu triple action powder - analgesic and antipyretic decongestant - 10 sachets
Estimated delivery: 1-3 business days
Information on returns and shipments
Payment methods
Medicines authorized by the Ministry of Health

Vicks
Sachets
Composition:
One sachet contains: 500 mg of paracetamol 200 mg of guaifenesin 10 mg of phenylephrine hydrochloride Excipients: Sucrose 2000 mg Aspartame 6 mg Sodium 157 mg For the full list of excipients, see section 6.1.
Excipients
Sucrose Citric acid Tartaric acid Sodium cyclamate Sodium citrate Aspartame (E951) Acesulfame potassium (E950) Menthol powder Lemon flavouring Lemon juice flavouring Quinoline yellow (E104)
Therapeutic indications
Short-term symptomatic treatment of mild to moderate pain, fever, nasal congestion with expectorant effect in case of productive cough, associated with colds, chills and flu.
Contraindications
Hypersensitivity to paracetamol, guaifenesin, phenylephrine hydrochloride or to any of the excipients. Hepatic impairment or severe renal impairment. Hypertension. Hyperthyroidism. Diabetes. Cardiac disease. Closed-angle glaucoma. Porphyria. Use in patients taking tricyclic antidepressants. Use in patients taking, or who have taken in the previous 2 weeks, monoamine oxidase inhibitors (MAOIs). Use in patients taking beta-blockers. Use in patients taking other sympathomimetic drugs. Children under 12 years of age.
Dosage
Dissolve the contents of one sachet in a medium-sized cup and add hot, not boiling, water (approximately 250 ml). Allow to cool to a drinkable temperature. Adults and children aged 12 years and over: one sachet. Repeat every four hours as directed, without exceeding four doses (sachets) in a 24-hour period. Do not administer to children under 12 years of age without medical advice. Do not administer to patients with hepatic impairment or severe renal impairment (see section 4.3). Consult your doctor if symptoms persist for more than 3 days.
Warnings and precautions
Prolonged use of the product is not recommended. Patients should be advised not to take other products containing paracetamol or containing the same active ingredients as this preparation. Patients should also be advised to avoid the concomitant use of alcohol, other decongestant products, or cough or cold products. The doctor or pharmacist is required to ensure that preparations containing sympathomimetics are not administered simultaneously through multiple routes, i.e., orally and topically (nasal, auricular, and ocular preparations). This medicine should only be recommended in the presence of all symptoms (pain and/or fever, nasal congestion, and bronchial cough). The risks associated with overdose are greater in patients with non-cirrhotic alcoholic liver disease. Use with caution in patients taking digitalis, beta-adrenergic blockers, methyldopa, or other antihypertensive agents (see section 4.5). Use with caution in patients with prostatic hypertrophy as they may be subject to urinary retention. Products containing sympathomimetics should be used with great caution in patients taking phenothiazines. Use in patients with Raynaud's phenomenon. Consult your doctor before use in case of persistent or chronic cough such as that caused by smoking, asthma, chronic bronchitis, or emphysema. Contains sucrose. Patients with rare hereditary problems of fructose intolerance, glucose-galactose malabsorption, or sucrase-isomaltase insufficiency should not take this medicine. Contains 157 mg sodium per dose. To be taken into consideration by patients with reduced kidney function or on a controlled sodium diet. Contains aspartame (E951). This medicine contains a source of phenylalanine. May be harmful if you have phenylketonuria.
Interactions
The hepatotoxicity of paracetamol may be enhanced by excessive alcohol intake. The rate of paracetamol absorption may be increased with metoclopramide or domperidone, and absorption reduced with cholestyramine. Substances that induce hepatic microsomal enzymes, such as alcohol, barbiturates, monoamine oxidase inhibitors, and tricyclic antidepressants, may increase the hepatotoxicity of paracetamol, particularly following overdose. Isoniazid reduces the elimination of paracetamol, possibly potentiating its activity and/or toxicity, by inhibiting its metabolism in the liver. Probenecid halves the elimination of paracetamol by inhibiting its binding to glucuronic acid. A reduction in paracetamol should be considered if probenecid is being taken. Regular use of paracetamol may reduce the metabolism of zidovudine (increasing the risk of neutropenia). Interactions between sympathomimetic amines, such as phenylephrine, and monoamine oxidase inhibitors cause hypertensive effects. Phenylephrine may interact negatively with sympathomimetic agents and may reduce the effectiveness of beta-blockers, methyldopa, and other antihypertensive drugs (see section 4.4). The conditions in which these drugs are taken constitute contraindications to the use of the product. The anticoagulant effect of warfarin and other coumarin drugs may be enhanced by regular and prolonged use of paracetamol, with an increased risk of haemorrhages; occasional doses have no significant effects. Drug interactions between paracetamol and several other drugs have been reported. These interactions are considered unlikely to be clinically significant with temporary use and according to the recommended dosage regimen. Salicylates/aspirin may prolong the elimination half-life (t ½) of paracetamol. Paracetamol may reduce the bioequivalence of lamotrigine, possibly decreasing its effect due to increased metabolism in the liver. Digitalis may sensitize the myocardium to effects similar to sympathomimetic substances. Paracetamol may alter the phosphotungstate uric acid test and the blood glucose test.
Undesirable effects
The incidence of adverse effects is usually classified as follows: Very common (>10) Common (>1/100 to <1/10) Uncommon (>1/1,000 to <1/100) Rare (>1/10,000 to < 1/1,000) Very rare (<1/10,000) Not known (incidence cannot be classified based on the available data) Cardiac disorders: Phenylephrine may rarely be associated with tachycardia (from
Price Trend
This product has been on sale since 25/09/2017
In the last 30 days, the product's lowest price was 6,21 €