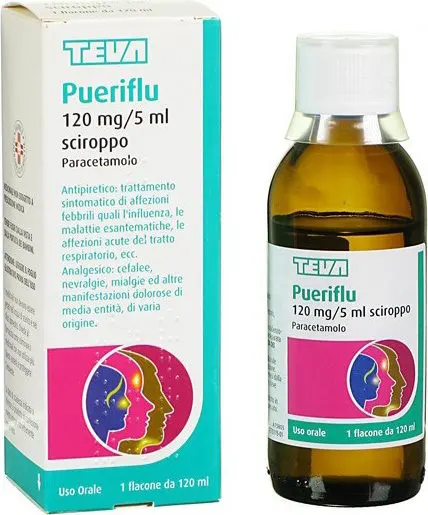
Pueriflu 120 mg/5 ml - sciroppo analgesico ed antipiretico 120 ml
Information on returns and shipments
Payment methods
Medicines authorized by the Ministry of Health

Syrup
Excipients
120 mg/5 ml syrup - 120 ml, 200 ml and 240 ml bottles Sucrose, macrogol 6000, sodium citrate, anhydrous citric acid, sodium saccharin, methyl p-hydroxybenzoate (E218), propyl p-hydroxybenzoate (E216), strawberry/mandarin flavouring, purified water.
120 mg/5 ml syrup - 10 single-dose containers of 5 and 10 ml Sucrose, macrogol 6000, sodium citrate, anhydrous citric acid, sodium saccharin, methyl p-hydroxybenzoate (E218), propyl p-hydroxybenzoate (E216), flavouring strawberry/mandarin, purified water.
COMPOSITION
120 mg/5 ml syrup
100 ml of syrup contain: paracetamol 2.4 g.
For the full list of excipients, see section 6.1.
PHARMACEUTICAL FORM
Syrup. Clear, colorless or slightly yellowish solution
Therapeutic indications
Dosage and method of administration
Contraindications
the excipients listed in paragraph 6.1
Warnings and precautions for use
In rare cases of allergic reactions, administration must be suspended and appropriate treatment must be initiated.
High or prolonged doses of the product can cause high-risk liver disease and alterations to the kidneys and blood, which may be serious.
Paracetamol should be administered with caution to patients with mild to moderate hepatocellular insufficiency (including Gilbert's syndrome), severe hepatic insufficiency (Child-Pugh >9), hepatitis B. acute, in concomitant treatment with drugs that alter liver function, glucose-6-phosphate dehydrogenase deficiency, haemolytic anaemia, renal failure. Therefore, administration should be carried out only if really necessary and under direct medical supervision. During treatment with paracetamol, before taking any other drug, check that it does not contain the same active ingredient, since if paracetamol is taken in high doses, serious adverse reactions can occur.
Instruct the patient to contact the doctor before combining any other drug. See also section 4.5.
PUERIFLU contains sucrose, which should be taken into account in diabetic patients and in patients on low-calorie diets, and methyl parahydroxybenzoate and propyl parahydroxybenzoate which can cause allergic reactions (even delayed). Patients with rare hereditary problems of fructose intolerance, glucose-galactose malabsorption, or sucrase-isomaltase insufficiency should not take this medicine.
Do not administer for more than 3 consecutive days without consulting your doctor.
Interactions
The oral absorption of paracetamol depends on the rate of gastric emptying. Therefore, concomitant administration of drugs that slow (e.g. anticholinergics, opioids) or increase (e.g. prokinetics) the rate of gastric emptying may result in a decrease or increase in the bioavailability of the product, respectively.
Concomitant administration of cholestyramine reduces the absorption of paracetamol. Concomitant use of paracetamol and chloramphenicol may increase the half-life of chloramphenicol, with the risk of increasing its toxicity.
Regular and prolonged use of paracetamol increases the effect of anticoagulants, in particular the concomitant use of paracetamol (4 g per day for at least 4 days) with oral anticoagulants may induce slight variations in INR values. In these cases, more frequent monitoring of INR values must be conducted during concomitant use and after its discontinuation.
Use Pueriflu with extreme caution and only under strict medical supervision during chronic treatment with drugs that can cause the induction of hepatic monooxygenases or in case of exposure to substances that can have this effect (for example, rifampicin,
cimetidine, antiepileptics such as glutethimide, phenobarbital, carbamazepine). The same applies in cases of alcoholism and in patients treated with zidovudine.
The administration of paracetamol may interfere with the determination of uric acid (using the phosphotungstic acid method) and blood glucose (using the glucose-oxidase-peroxidase method).
Pregnancy and breastfeeding
Although clinical studies in pregnant or breastfeeding patients have not highlighted any particular contraindications to the use of paracetamol or caused any adverse effects on the mother or child, it is recommended to administer the product only in cases of actual need and under the direct supervision of a doctor.
Side effects
With the use of paracetamol, skin reactions of various types and severity have been reported, including cases of erythema multiforme, Stevens-Johnson syndrome, and epidermal necrolysis.
Hypersensitivity reactions such as skin rashes with erythema or urticaria, angioedema, laryngeal edema, anaphylactic shock have been reported. In addition, the following adverse reactions have been reported: leukopenia, anaemia, liver function disorders and hepatitis, renal disorders (acute renal failure, interstitial nephritis, haematuria, anuria), gastrointestinal reactions and dizziness.
Overdose
In case of accidental ingestion of very high doses of paracetamol, acute intoxication manifests itself with anorexia, nausea, and vomiting followed by a profound deterioration in general conditions; these symptoms generally appear within the first 24 hours. In case of overdose, paracetamol can cause necrosis of the renal tubules and hepatic cytolysis, which can progress to massive and irreversible necrosis, resulting in hepatocellular failure, metabolic acidosis, and encephalopathy, which can lead to coma and death. Simultaneously, an increase in liver transaminase, lactate dehydrogenase, and bilirubin levels, and a reduction in prothrombin levels are observed, which may occur within 12-48 hours of ingestion.
The measures to be taken consist of hospitalization for appropriate treatment, with the administration, as soon as possible, of N-acetylcysteine as an antidote: the dosage is 150 mg/kg IV in glucose solution over 15 minutes, then 50 mg/kg over the next 4 hours and 100 mg/kg over the next 16 hours, for a total of 300 mg/kg over 20 hours.
Price Trend
This product has been on sale since 25/09/2017
In the last 30 days, the product's lowest price was 2,74 €