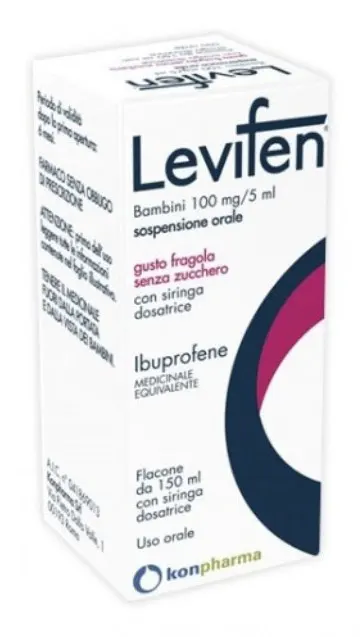
Levifen 100mg/5ml strawberry - children healt medicament 150ml
Information on returns and shipments
Payment methods
Medicines authorized by the Ministry of Health

Medicine indicated for the symptomatic treatment of fever and mild to moderate pain.
Indications
Levifen Children contains ibuprofen, a nonsteroidal anti-inflammatory drug (NSAID) with analgesic (pain-relieving), antipyretic (fever-reducing) and anti-inflammatory properties.
- Strawberry Flavor
- Sugar-free
Composition
Active ingredients
The active ingredient is ibuprofen. Each ml of suspension contains 20 mg of ibuprofen.
Excipients
The other ingredients are: citric acid monohydrate, sodium citrate, acesulfame potassium, xanthan gum, sodium benzoate, strawberry flavouring, maltitol syrup, glycerin, purified water.
How to use and Dosage
Always take this medicine exactly as described in this leaflet or as your doctor or pharmacist has told you. If you are not sure, check with your doctor or pharmacist.
The recommended dose depends on the weight and age of the child. It is recommended to limit the administration of this medicine to children over 3 months of age and weighing more than 5.6 kg.
Undesirable effects may be minimised by using the lowest effective dose for the shortest duration necessary to control symptoms. Oral administration to infants and children aged between 3 months and 12 years should be done using the dosing syringe found in the medicine package. The graduated scale on the syringe clearly shows the markings for the different dosages; in particular, the 2.5 ml mark (corresponding to 50 mg of ibuprofen) and the 5 ml mark (corresponding to 100 mg of ibuprofen). The recommended daily dose of 20-30 mg of medicine per kg of the child's body weight, to be divided into 3 administrations per day to be taken at intervals of 6-8 hours, can be administered according to the following schedule.
- Children aged between 3 and 6 months (5.6 - 7 kg of body weight) and children aged between 6 and 12 months (7-10 kg of body weight). The recommended dose is 2.5 ml taken 3 times a day at intervals of 6-8 hours.
- Children aged 1 to 3 years (10-15 kg body weight). The recommended dose is 5 ml taken 3 times a day at intervals of 6-8 hours.
- Children aged 4 to 6 years (15-20 kg body weight). The recommended dose is 7.5 ml (5 ml + 2.5 ml) taken 3 times a day at intervals of 6-8 hours.
- Children aged 7 to 9 years (20-28 kg body weight). The recommended dose is 10 ml (5 ml + 5 ml) to be taken 3 times a day at intervals of 6-8 hours.
- Children aged between 10 and 12 years (28-43 kg body weight). The recommended dose is 15 ml to be taken 3 times a day at intervals of 6-8 hours.
In case of fever following vaccination, refer to the dosage indicated above, administering a single dose followed, if necessary, by another dose after 6 hours. Do not administer more than two doses in 24 hours. Consult your doctor if the fever does not decrease. In children aged between 3 and 5 months, consult your doctor if the symptoms persist for a period of more than 24 hours or if the symptoms worsen. If use of the medicine is necessary for more than 24 hours, consult your doctor. 3 days in children aged between 3 and 5 months and in children over 6 months, or in case of worsening of the symptoms, a doctor must be consulted.
Instructions for using the dosing syringe:
- Unscrew the cap by pushing it downwards and turning it to the left.
- Insert the tip of the syringe fully into the hole in the undercap.
- Shake well.
- Invert the bottle, then, holding the syringe firmly, gently pull the plunger downwards, allowing the suspension to flow into the syringe until it reaches the mark printed on the plunger corresponding to the desired dose.
- Return the bottle to an upright position and remove the syringe by gently rotating it.
- Insert the tip of the syringe into the child's mouth and apply gentle pressure on the plunger to allow the suspension to flow out.
- After use, screw the cap to close the bottle and rinse the syringe with hot water. Leave to dry, keeping it out of the reach and sight of children.
Warnings
Talk to your doctor or pharmacist before using Levifen.
Undesirable effects can be minimized by using the lowest effective dose for the shortest duration of treatment necessary to control symptoms. Analgesics, antipyretics, and non-steroidal anti-inflammatory drugs can cause allergic reactions (hypersensitivity), even in people who have not previously taken this type of drug. The risk of allergic reactions after taking ibuprofen is higher in people who have experienced such reactions after using other analgesics, antipyretics, and nonsteroidal anti-inflammatory drugs, and in people with asthma, nasal problems (polyposis), or who have had allergic reactions in the past that manifested as facial swelling (angioedema). Allergic reactions occur especially in the initial phases of treatment (first month). Serious skin reactions, some of them fatal, including exfoliative dermatitis, Stevens-Johnson syndrome, and toxic epidermal necrolysis, have been reported rarely with the use of anti-inflammatory drugs.
Stop taking Levifen Paediatric at the first appearance of skin rash (eruption), mucosal lesions, or any other sign of an allergic reaction.
Stomach and/or intestinal problems
During treatment with anti-inflammatory drugs (NSAIDs), stomach bleeding (gastrointestinal bleeding, ulceration, and perforation), which can be fatal, may occur. These events can occur at any time, with or without warning symptoms or a previous history of serious gastrointestinal events. Elderly patients have an increased frequency of side effects to NSAIDs, especially gastrointestinal bleeding and perforation, which can be fatal. In the elderly and in patients with a history of ulcers, especially if complicated by haemorrhage or perforation, the risk of gastrointestinal bleeding, ulceration, or perforation is higher when using high doses of NSAIDs. If you belong to this category of patients, you should start treatment with the lowest available dose. In these cases, talk to your doctor, who may advise you to combine Levifen with stomach-protective medicines, especially if you take other medicines (for example, aspirin or other medicines that increase the risk of gastrointestinal problems). During treatment with this medicine, especially if you are elderly or have a history of stomach or intestinal problems, you should report any unusual symptoms (especially gastrointestinal bleeding) to your doctor, particularly in the initial stages of treatment. If bleeding or ulcers in the stomach and/or intestines occur, stop taking Levifen Paediatrics.
Use this medicine with caution and contact your doctor to discuss treatment with Levifen if:
- you are taking medicines that may increase the risk of ulceration or bleeding, such as oral corticosteroids (cortisone and similar), anticoagulants such as warfarin (used to thin the blood), selective serotonin reuptake inhibitors (used as antidepressants) or antiplatelet agents such as aspirin (which block platelets, the cells responsible for clotting);
- you have suffered in the past from intestinal disorders (ulcerative colitis, Crohn's disease, inflammatory bowel disease);
- you have suffered or suffer from high blood pressure (hypertension);
- you suffer from heart problems (heart failure, heart failure congestive heart disease, ischemic heart disease);
- have or have had peripheral arterial disease (poor circulation in the legs or feet due to narrowing or blockage of the arteries) or have had any type of stroke (including 'mini-stroke' or 'TIA', transient ischemic attack);
- have breathing problems (asthma);
- have blood clotting disorders;
- have kidney, heart or liver disease;
- have risk factors for cardiovascular events such as high blood pressure, increased fats in the blood (hyperlipidaemia), diabetes mellitus and smoking.
The use of ibuprofen, particularly at high doses and in long-term treatment, may be associated with a small increased risk of heart attack or stroke (arterial thrombotic events). Do not exceed the recommended dose or duration of treatment. Avoid using Levifen if you are taking other similar medicines (nonsteroidal anti-inflammatory drugs, NSAIDs, including selective COX-2 inhibitors), as the risk of side effects may increase. The use of Levifen Children, acetylsalicylic acid or other analgesics, antipyretics, non-steroidal anti-inflammatory drugs requires particular caution:
- in case of previous gastrointestinal ulcerations, perforations or bleeding;
- in case of asthma;
- in the presence of coagulation defects (reduced ability of the blood to clot);
- in the presence of kidney, heart disease or high blood pressure, especially if you are an elderly patient or if you have impaired kidney or liver function, have heart failure or are being treated with diuretics, drugs used in the treatment of high blood pressure and heart failure);
- in the presence of liver disease;
- in case of dehydration (for example due to fever, vomiting or diarrhea). You will need to be rehydrated before starting and during treatment.
During treatment, especially if prolonged:
- Look out for signs or symptoms of ulceration or bleeding in the stomach and intestines and report them immediately to your doctor;
- Look out for signs or symptoms of toxicity to the liver or kidneys and report them immediately to your doctor;
- If you experience visual disturbances (blurred or reduced vision, scotomas, i.e. loss of vision in some parts of the visual field, alterations in colour perception), stop treatment and consult your doctor;
- If you experience signs or symptoms of meningitis (inflammation of the membranes covering the brain): contact your doctor who will have to evaluate the rare possibility that it is due to the use of this medicine (more frequent in subjects suffering from systemic lupus erythematosus or other collagen diseases).
Children
There is a risk of impaired renal function in dehydrated children. Levifen Children contains:
- Maltitol. If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicine;
- less than 1 mmol (23 mg) sodium per dose (2.5 ml), that is to say essentially 'sodium-free'.
- Levifen Bambini does not contain sugar and is therefore indicated in patients who need to control their sugar and calorie intake.
Contraindications
Do not take Levifen Bambini
- If you are allergic to ibuprofen or any of the other ingredients of this medicine;
- If you are allergic to acetylsalicylic acid and other painkillers (analgesics), fever reducers (antipyretics), non-steroidal anti-inflammatory drugs (NSAIDs), in particular when the allergy is associated with nasal congestion due to secretion of the mucous membrane inside the nose (nasal polyposis) and asthma;
- If you suffer from an active peptic ulcer (ulcer of the stomach and first part of the intestine);
- In case of children under 3 months of age or weighing less than 5.6 kg;
- If you suffer from severe kidney or liver problems (severe renal or hepatic insufficiency);
- If you suffer from severe heart problems (severe cardiac insufficiency);
- If you are pregnant or breastfeeding;
- If you have suffered in the past from stomach or intestinal problems associated with bleeding or lesions (gastrointestinal bleeding or perforation related to previous active treatments or history of recurrent peptic bleeding/ulcer);
- If you are taking other anti-inflammatory medicines (NSAIDs, including selective COX-2 inhibitors) at the same time.
Interactions with other medicines
Tell your doctor or pharmacist if you are taking, have recently taken taken or might take any other medicines. Consult your doctor before using Levifen with other medicines, in particular:
- avoid the simultaneous use of two or more analgesics, antipyretics, non-steroidal anti-inflammatory drugs: as it increases the risk of side effects;
- corticosteroids (cortisone and similar), medicines used to reduce inflammation and to treat allergies, as it increases the risk of gastrointestinal ulceration or bleeding;
- antibacterials, as it increases the risk of gastrointestinal ulceration or bleeding; there is a possible increased risk of seizures;
- anticoagulants such as warfarin (used to thin the blood);
- antiplatelet agents and selective serotonin reuptake inhibitors (SSRIs) (drugs used to treat depression);
- antidiabetics;
- antivirals (ritonavir);
- ciclosporin, a drug used to prevent organ transplant rejection;
- cytotoxic anticancer drugs (methotrexate);
- lithium, a medicine used in some psychiatric illnesses;
- tacrolimus, a drug used to prevent organ transplant rejection;
- probenecid, a medicine used to lower the level of uric acid in the blood;
- methotrexate, an immune system suppressant;
- zidovudine, used in HIV infection;
- diuretics, ACE inhibitors, and angiotensin II antagonists (drugs used to lower blood pressure).
During treatment with Levifen in combination with other medicines, it is important that you drink adequate fluids and have regular checks of your kidney function. If you are being treated with acetylsalicylic acid (aspirin), consult your doctor before taking Levifen. Ibuprofen may inhibit the effects of acetylsalicylic acid on platelet aggregation.
Side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them. The following serious side effects may occur with the use of Levifen Children:
- Gastrointestinal effects, such as: peptic ulcers (of the stomach and upper intestine), perforations or gastrointestinal bleeding, sometimes fatal, particularly in the elderly
- Allergic reactions, hives with or without swelling of the face (angioedema), shock, syndrome characterized by abdominal pain, fever, chills, nausea and vomiting, difficulty breathing, bronchospasm (closure of the bronchi).
- Hepatitis (inflammation of the liver), jaundice (yellowing of the skin), alteration of liver function tests, pancreatitis (inflammation of the pancreas), duodenitis (inflammation of a part of the intestine), esophagitis (inflammation of the esophagus, the tube that connects the mouth to the stomach), hepatorenal syndrome (a complication of liver disease, with involvement of the kidney), liver necrosis (death of liver cells), liver failure. liver.
- Nausea, vomiting, diarrhoea, flatulence (passing of gas from the intestines), constipation (constipation), dyspepsia (indigestion), abdominal pain, melaena (presence of digested blood in the stools), haematemesis (vomiting blood), ulcerative stomatitis (mouth ulcers), worsening of colitis and Crohn's disease (inflammatory bowel disease);
- Stomach pain, gastritis, heartburn (heartburn). Stomach problems may be reduced by taking the medicine on a full stomach.
- Dizziness, headache, irritability, tinnitus (perception of a ringing in the ear).
- Skin rashes (including maculopapular) itching.
- Decreased appetite.
- Swelling due to accumulation of fluid in a part of the body (edema).
- High blood pressure and heart failure.
- Fluid retention (usually responds promptly to discontinuation of treatment).
- In patients with pre-existing autoimmune diseases (e.g. systemic lupus erythematosus, connective tissue diseases), individual cases of symptoms of aseptic meningitis such as neck tension, headache, nausea, vomiting, fever, disorientation have been reported.
- Depression, insomnia, difficulty concentrating, emotional instability, drowsiness, aseptic meningitis, convulsions, hearing and vision disorders.
- Bronchospasm (closure of the bronchi), dyspnoea (difficulty breathing), apnoea (stopping of breathing).
- Skin disorders with the formation of blisters and bullae (vesiculobullous eruptions), urticaria, skin irritation (erythema multiforme, exfoliative dermatitis, photosensitivity dermatitis).
- Severe skin disorders (bullous reactions including Stevens-Johnson syndrome and toxic epidermal necrolysis).
- Alopecia (hair loss).
- Dry eyes and mouth, gum ulcers, rhinitis (inflammation of the nasal mucosa).
- Gastritis.
- Miscellaneous Blood problems: neutropenia (decrease in neutrophils, a type of white blood cell). agranulocytosis (severe reduction in white blood cells), aplastic anemia (anemia caused by poor bone marrow function), haemolytic anemia (anemia caused by the breakdown of white blood cells) (possible positive Coombs test, a test that checks the compatibility of blood), thrombocytopenia (decrease in the number of platelets, the cells responsible for clotting) (with or without purpura, small bleeding in the skin), eosinophilia (increase in the number of eosinophils, a type of white blood cell), reduction in haemoglobin and haematocrit (degree of concentration of blood), pancytopenia (decrease in all types of blood cells).
- Cerebrovascular accidents (such as stroke), low blood pressure, heart problems (congestive heart failure in people with impaired heart function).
- Palpitations.
- Kidney problems (acute renal failure in people with existing renal impairment, necrosis papillary effusion, tubular necrosis, glomerular nephritis, alteration of renal function tests). Polyuria (passage of large amounts of urine), cystitis (inflammation of the bladder), hematuria (blood in the urine).
The use of ibuprofen, especially at high doses (2400 mg/day) and for long-term treatment, may be associated with a small increased risk of arterial thrombotic events (e.g. myocardial infarction or stroke). A serious skin reaction known as DRESS syndrome may occur. Symptoms of DRESS include: rash, fever, swollen lymph nodes and an increase in eosinophils (a type of white blood cell).
Overdose
If you take too much ibuprofen (overdose), you may experience more serious side effects. The following symptoms commonly occur: nausea, vomiting, stomach pain, deep sleep with reduced response to normal stimuli (lethargy), drowsiness, headache, dizziness, ringing in the ears (tinnitus), uncontrolled body movements (convulsions), and loss of consciousness. Symptoms of an overdose may appear within 4–6 hours of taking ibuprofen. Rarely, it may cause: uncontrolled eye movements (nystagmus), increased blood acid levels (metabolic acidosis), low body temperature (hypothermia), kidney effects, bleeding in the stomach and intestines, profound loss of consciousness (coma), temporary cessation of breathing (apnea), diarrhea, reduced nervous system activity (Central Nervous System depression) and respiratory activity (respiratory system depression). In addition, it may cause: You may also experience: disorientation, excitement, fainting, low blood pressure (hypotension), slow or fast heartbeat (bradycardia or tachycardia). If you take significantly high doses of ibuprofen, you may experience serious damage to your kidneys and liver. If you have taken more Levifen CHILDREN than you should, or if your child has taken this medicine by mistake, always contact a doctor or the nearest hospital to receive an opinion on the risk and advice on what action to take. Symptoms may include nausea, stomach pain, vomiting (which may contain traces of blood), headache, ringing in the ears, confusion, and uncontrolled eye movements. At high doses, drowsiness, chest pain, palpitations, loss of consciousness, convulsions (especially in children), weakness and dizziness, blood in the urine, feeling cold, and breathing problems have been reported. If you have any further questions on the use of Levifen, ask your doctor or pharmacist.
Pregnancy and breastfeeding
If you are pregnant or breastfeeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine. Children under 12 years of age are unlikely to become pregnant or breastfeed. However, in these cases, it should be borne in mind that taking Levifen can have serious and harmful effects on the fetus. This medicine should not be used during pregnancy or breastfeeding.
Storage
Keep this medicine out of the sight and reach of children. Do not use this medicine after the expiry date which is stated on the carton after EXP. The expiry date refers to the last day of that month and to the product in an intact package, stored correctly. Shelf life after first opening: 6 months.
Format
Oral suspension, 150 ml bottle with dosing syringe.
Price Trend
This product has been on sale since 09/03/2023
In the last 30 days, the product's lowest price was 10,54 €