Aspirina dolore infiammazione 500 mg 20 tablets
Estimated delivery: 1-3 business days
-
20 tablets€7.48
-
8 tablets€4.78
Information on returns and shipments
Payment methods
Medicines authorized by the Ministry of Health

Active ingredients
Each tablet contains 500 mg of acetylsalicylic acid.
Excipients
Tablet core: Colloidal silicon dioxide, Anhydrous sodium carbonate
Coating: Carnauba wax, Hypromellose, Zinc stearate.
Therapeutic indications
Symptomatic treatment of fever and/or mild to moderate pain, such as headache, flu-like syndrome, toothache, muscle pain.
Contraindications
• Hypersensitivity to acetylsalicylic acid or other salicylates, or to any of the excipients.
• history of asthma or hypersensitivity reactions (e.g. urticaria, angioedema, severe rhinitis, shock) induced by the administration of salicylates or substances with a similar action, in particular nonsteroidal anti-inflammatory drugs (NSAIDs)
• active peptic ulcer
• haemorrhagic diathesis
• severe renal insufficiency
• severe hepatic insufficiency
• severe uncontrolled cardiac insufficiency
• concomitant administration of methotrexate in doses exceeding 20 mg per week, for anti-inflammatory doses of acetylsalicylic acid, or for analgesic or antipyretic doses
• concomitant administration of oral anticoagulants for anti-inflammatory doses of acetylsalicylic acid, or for analgesic or antipyretic doses and in patients with a history of gastroduodenal ulcers.
• from the beginning of the 6th month of pregnancy (beyond the twenty-fourth week of amenorrhea).
• children and adolescents under 16 years of age.
Dosage
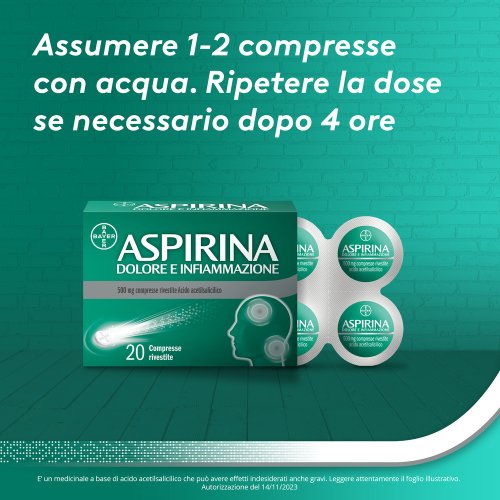
Dosage Adults and adolescents (16 years and older): 1 to 2 tablets for each dose to be repeated as needed after a minimum period of 4 hours. The maximum daily dose must not exceed 6 tablets. Elderly (from 65 years): 1 tablet for each dose to be repeated as needed after a minimum period of 4 hours. The maximum daily dose must not exceed 4 tablets. Acetylsalicylic acid should not be taken for more than 12 hours. 3 days (in case of fever) or 3–4 days (in case of pain) unless otherwise indicated by your doctor. Paediatric population: Acetylsalicylic acid should not be used in children and adolescents under 16 years of age without a medical prescription. Acetylsalicylic acid should be used with caution in patients with abnormal liver or kidney function or with circulatory problems.
Method of administration
For oral use. The tablets should be taken with an adequate amount of liquid.
Storage
Do not store above 30°C.
Warnings
In case of combination with other medicinal products, to avoid any risk of overdose, check that acetylsalicylic acid is not in the composition of these other medicines. • Reye's syndrome, a very rare and potentially fatal disease, has been described in children with symptoms of viral infections (particularly chickenpox and influenza) with or without aspirin intake. Therefore, aspirin should be administered to children with these conditions only after medical advice and when other measures have proven ineffective. If persistent vomiting, altered consciousness, or abnormal behavior occurs, aspirin treatment should be discontinued. If high-dose analgesics are administered for a long time, headache attacks should not be treated with higher doses. Regular use of analgesics, especially combinations of analgesics, can lead to permanent kidney damage, with the risk of kidney failure. In some severe forms of G6PD deficiency, high doses of aspirin can cause hemolysis. In case of G6PD deficiency, acetylsalicylic acid should be administered under medical supervision. Treatment monitoring should be intensified in the following cases: in patients with a history of gastric or duodenal ulcer, gastrointestinal bleeding, or gastritis. in patients with renal insufficiency; in patients with hepatic insufficiency; in patients with asthma: the occurrence of an asthma attack, in some patients, may be linked to an allergy to nonsteroidal anti-inflammatory drugs or acetylsalicylic acid; in this case, this medicine is contraindicated. in patients with metrorrhagia or menorrhagia (risk of increased volume and duration of the cycle). Gastrointestinal bleeding or ulcers/perforations can occur at any time during treatment, without necessarily having any warning signs or medical history in the patient. The relative risk increases in elderly subjects, in subjects with low body weight, and in patients receiving anticoagulants or platelet aggregation inhibitors. In case of gastrointestinal bleeding, treatment must be discontinued immediately. Given the inhibitory effect of acetylsalicylic acid on platelet aggregation, which occurs even at very low doses and persists for several days, the patient should be aware of the risk of haemorrhage in case of surgical interventions, even minor ones (e.g. tooth extraction). In analgesic or antipyretic doses, acetylsalicylic acid inhibits the excretion of uric acid; in doses used in rheumatology (anti-inflammatory doses), acetylsalicylic acid has a uricosuric effect. The use of this medicine is not recommended during breastfeeding. The administration of acetylsalicylic acid is not recommended with: Oral anticoagulants with analgesic or antipyretic doses of acetylsalicylic acid (≥500 mg per administration and/or < 3 g per day) and in patients without a history of gastro-duodenal ulcers • Other nonsteroidal anti-inflammatory drugs (NSAIDs) with anti-inflammatory doses of acetylsalicylic acid (≥ 1 g per administration and/or ≥ 3 g per day) or with analgesic or antipyretic doses of acetylsalicylic acid (≥ 500 mg per administration and/or < 3 g per day) • Low molecular weight heparins (and related molecules) and unfractionated heparins at therapeutic doses or in elderly patients (> 65 years) regardless of the heparin dose, and for anti-inflammatory doses of acetylsalicylic acid (> 1 g per administration and/or > 3 g per day) or with analgesic or antipyretic doses of acetylsalicylic acid (> 500 mg per administration and/or < 3 g per day) • Clopidogrel (beyond the approved indications for this combination in patients with acute coronary disease) • Ticlopidine • Uricosurics • Glucocorticoids (except hydrocortisone replacement therapy) at anti-inflammatory doses of acetylsalicylic acid (> 1 g per administration and/or > 3 g per day) • Pemetrexed in patients with mild to moderately reduced renal function (creatinine clearance between 45 ml/min and 80 ml/min) • Anagrelide: increased risk of haemorrhage and decreased antithrombotic effect
Interactions
In the following text, the following definitions apply: – anti-inflammatory doses of acetylsalicylic acid are defined as "≥ 1g per administration and/or ≥ 3g per day". – Analgesic or antipyretic doses of acetylsalicylic acid are defined as "≥500 mg per administration and/or <3 g per day". Several substances give rise to interactions due to their properties as inhibitors of platelet aggregation: abciximab, acetylsalicylic acid, cilostazol, clopidogrel, epoprostenol, eptifibatide, iloprost, iloprost trometamol, prasugrel, ticlopidine, tirofiban, ticagrelor. The risk of bleeding increases with the use of multiple platelet aggregation inhibitors as well as with their use in combination with heparin or related molecules, oral anticoagulants or other thrombolytics, and must be assessed through constant clinical monitoring. Contraindicated combinations • Methotrexate in doses greater than 20 mg per week, with anti-inflammatory doses of acetylsalicylic acid, or with analgesic or antipyretic doses of acetylsalicylic acid: Increased toxicity of methotrexate, particularly haematological toxicity (due to reduced renal elimination of methotrexate caused by acetylsalicylic acid). Oral anticoagulants with anti-inflammatory doses of acetylsalicylic acid, or with analgesic or antipyretic doses of acetylsalicylic acid and in patients with a history of gastroduodenal ulcers: Increased risk of haemorrhage. Combinations not recommended: Oral anticoagulants with analgesic or antipyretic doses of acetylsalicylic acid and in patients without a history of gastroduodenal ulcers: Increased risk of haemorrhage. Other nonsteroidal anti-inflammatory drugs (NSAIDs) with anti-inflammatory doses of acetylsalicylic acid, or with analgesic or antipyretic doses of acetylsalicylic acid: increased risk of gastrointestinal ulcers and haemorrhage. • Low molecular weight heparins (and related molecules) and unfractionated heparins at curative doses, or in elderly patients (>65 years) regardless of the heparin dose, and for anti-inflammatory doses of acetylsalicylic acid or analgesic or antipyretic doses of acetylsalicylic acid: increased risk of haemorrhage (inhibition of platelet aggregation and aggression of the gastroduodenal mucosa by acetylsalicylic acid). Another anti-inflammatory drug, or another analgesic or antipyretic, should be used. • Clopidogrel (outside the approved indication for this combination in patients with acute coronary syndrome): increased risk of haemorrhage. If concomitant administration cannot be avoided, clinical monitoring is recommended. Ticlopidine: Increased risk of haemorrhage. If concomitant administration cannot be avoided, clinical monitoring is recommended. Uricosurics (benzbromarone, probenecid): Reduced uricosuric effect due to competition for uric acid elimination in the renal tubules. Glucocorticoids (excluding hydrocortisone replacement therapy) at anti-inflammatory doses of acetylsalicylic acid: Increased risk of haemorrhage. Pemetrexed in patients with mild to moderate renal impairment (creatinine clearance between 45 ml/min and 80 ml/min); increased risk of pemetrexed toxicity (due to decreased renal elimination of pemetrexed caused by acetylsalicylic acid) at anti-inflammatory doses of acetylsalicylic acid. • Anagrelide: increased risk of haemorrhage and decreased antithrombotic effect. If concomitant administration cannot be avoided, clinical monitoring is recommended. Combinations requiring precautions for use: • Diuretics, angiotensin-converting enzyme (ACE) inhibitors, and angiotensin II receptor antagonists, with anti-inflammatory doses of acetylsalicylic acid or with analgesic or antipyretic doses of acetylsalicylic acid: Acute renal failure may occur in dehydrated patients due to a reduction in glomerular filtration rate caused by decreased renal prostaglandin synthesis. Furthermore, there may be a reduction in the antihypertensive effect. Ensure that the patient is hydrated and that renal function is monitored at the start of treatment. • Methotrexate in doses ≥ 100 mg. 20 mg weekly, with anti-inflammatory doses of acetylsalicylic acid, or with analgesic or antipyretic doses of acetylsalicylic acid: Increased methotrexate toxicity, particularly hematologic toxicity (due to reduced renal elimination of methotrexate caused by acetylsalicylic acid). Blood counts should be monitored weekly during the first weeks of concomitant administration. Patients with reduced renal function (even mild) and elderly patients should be closely monitored. Clopidogrel (in the approved indication for this combination in patients with acute coronary syndrome): Increased risk of bleeding. Clinical monitoring is recommended. Topical gastrointestinal treatments, antacids, and activated charcoal: Increased renal excretion of acetylsalicylic acid due to urine alkalinization. It is recommended that antacids and topical gastrointestinal treatments be administered at least two hours after acetylsalicylic acid. Pemetrexed in patients with normal renal function: Increased risk of pemetrexed toxicity (due to decreased renal elimination of pemetrexed caused by acetylsalicylic acid) with anti-inflammatory doses of acetylsalicylic acid. Renal function should be monitored. Combinations that should be considered: Glucocorticoids (excluding hydrocortisone replacement therapy) for analgesic and antipyretic doses of acetylsalicylic acid: Increased risk of haemorrhage. Deferasirox: With anti-inflammatory doses of acetylsalicylic acid, or with analgesic or antipyretic doses of acetylsalicylic acid: Increased risk of gastrointestinal ulcers and haemorrhage. Low molecular weight heparins (and related molecules) and unfractionated heparins in preventive doses in patients under 65 years of age: by affecting hemostasis to various degrees, concomitant administration increases the risk of bleeding. Therefore, in patients under 65 years of age, the concomitant administration of heparins (or related molecules) in preventive doses, and acetylsalicylic acid at any dose, should be considered in combination with clinical and laboratory monitoring as needed. Thrombolytics: increased risk of bleeding. Selective serotonin reuptake inhibitors (citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline): increased risk of bleeding. Side effects: Frequencies: Not known (cannot be estimated from the available data). Blood and lymphatic system disorders: Bleeding and tendency to bleed (epistaxis, bleeding gums, purpura, etc.) with increased bleeding time. The risk of bleeding may persist for 4–8 days after stopping acetylsalicylic acid. May cause an increased risk of haemorrhage during surgery. Intracranial and gastrointestinal haemorrhages may also occur. Immune system disorders: Hypersensitivity reactions, anaphylactic reactions, asthma, angioedema. Nervous system disorders: Headache, dizziness, sensation of hearing loss, tinnitus, usually indicating an overdose. Intracranial haemorrhage. Gastrointestinal disorders: Abdominal pain. Occult or overt gastrointestinal haemorrhage (haematemesis, melena, etc.) resulting in iron deficiency anaemia. The risk of bleeding is dose-related. Gastric ulcers and perforations Hepatobiliary disorders Elevation of liver enzymes usually reversible with discontinuation of treatment, liver damage, mainly of hepatocellular nature Skin and subcutaneous tissue disorders Urticaria, rash General disorders Reye's syndrome (Reporting of side effects It is important to report side effects of the medicinal product after authorisation. This allows for continued monitoring of the risk-benefit balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the website: www.agenziafarmaco.gov.it/it/responsabili. Overdose Overdose may be harmful in elderly subjects and particularly in young children (therapeutic overdose or, more frequently, accidental intoxication) in whom it can be fatal. Symptoms Moderate intoxication: Symptoms such as ringing in the ears, sensation of hearing loss, headache and dizziness are indicative of an overdose and can be kept under control. Control by dose reduction. Severe intoxication: Symptoms include: Fever, hyperventilation, ketosis, respiratory alkalosis, metabolic acidosis, coma, cardiovascular collapse, respiratory failure, severe hypoglycemia. In children, overdose can be fatal starting from a single dose of 100 mg/kg. Emergency management • Immediate transfer to a specialized hospital unit • Gastrointestinal lavage and administration of activated charcoal • Monitoring of acid-base balance • Alkalinization of urine with monitoring of urinary pH • Hemodialysis in case of severe intoxication • Symptomatic treatment
Pregnancy and breastfeeding
Pregnancy
Inhibition of prostaglandin synthesis may have adverse effects on the course of pregnancy and/or embryo-fetal development. Data from studies Epidemiological data suggest an increased risk of miscarriage, cardiac malformations, and gastroschisis following the use of prostaglandin synthesis inhibitors in early pregnancy. The absolute risk of cardiovascular malformations is increased from no less than 1% to approximately 1.5%. The risk appears to increase with dose and duration of treatment. In animals, administration of a prostaglandin synthesis inhibitor has been shown to result in increased pre- and post-implantation loss and embryo-fetal mortality. Furthermore, an increased incidence of various malformations, including cardiovascular ones, has been reported in animals administered a prostaglandin synthesis inhibitor during the organogenetic period of gestation. Unless absolutely essential, acetylsalicylic acid should not be administered during the first 24 weeks of amenorrhea. If acetylsalicylic acid is administered to women attempting to conceive or who are pregnant during the first 24 weeks of pregnancy, the following should be considered: After 24 weeks of amenorrhea, the dose should be as low as possible and the duration of treatment as short as possible. After the 24th week of amenorrhea, all prostaglandin synthesis inhibitors may expose the fetus to: • cardiopulmonary toxicity (with premature closure of the ductus arteriosus and pulmonary hypertension); • renal dysfunction, which may progress to renal failure with oligohydramnios; In the final phase of pregnancy, the mother and the newborn may experience: • Prolongation of bleeding time, due to the inhibition of platelet aggregation, which may occur even at very low doses of acetylsalicylic acid; • inhibition of uterine contractions, which causes a delay or prolongation of labor. Therefore, acetylsalicylic acid is contraindicated after the 5th month of pregnancy (after 24 weeks of amenorrhea). amenorrhea)
Breastfeeding
Acetylsalicylic acid passes into breast milk: therefore, the use of acetylsalicylic acid is not recommended during breastfeeding.
Fertility
There is some evidence that drugs that inhibit cyclooxygenase/prostaglandin synthesis may cause impairment of female fertility due to an effect on ovulation. This effect is reversible upon discontinuation of treatment.
Price Trend
This product has been on sale since 17/12/2018
In the last 30 days, the product's lowest price was 7,28 €