Free shipping from 69,00 €
Get free shipping with a minimum spend of 69,00 €. Or by purchasing one of the selected products. Discover them now!
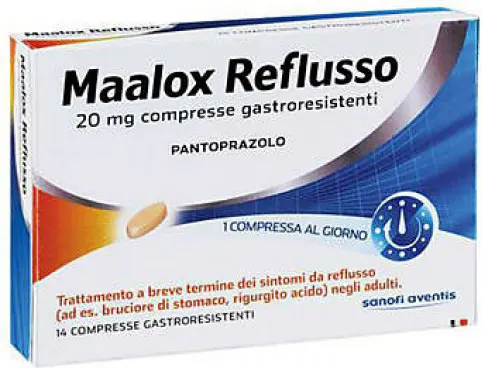
Maalox reflux 20 mg - treatment of gastroesophageal reflux - 14 gastro-resistant tablets
Information on returns and shipments
Payment methods
Medicines authorized by the Ministry of Health

Gastro-resistant tablets
Composition:
Each gastro-resistant tablet contains 20 mg pantoprazole (as sodium sesquihydrate). Excipients: 38.425 mg Maltitol and 0.345 mg Lecithin (derived from soya oil).
Excipients
Core maltitol (E965) crospovidone type B sodium carmellose sodium carbonate anhydrous calcium stearate Coating Poly(vinyl alcohol) talc titanium dioxide (E 171) macrogol 3350 soya lecithin yellow iron oxide (E 172) sodium carbonate anhydrous methacrylic acid copolymer ethyl acrylate (1:1) sodium lauryl sulphate polysorbate 80 triethyl citrate
Therapeutic indications: Short-term treatment of reflux symptoms (e.g. heartburn, acid regurgitation) in adults.
Contraindications: Hypersensitivity to the active substance, substituted benzimidazoles, lecithin (derived from soybean oil) or to any of the excipients. Concomitant administration with atazanavir.
Dosage
Dosage The recommended dose is 20 mg pantoprazole (one tablet) daily. It may be necessary to take the tablets for 2-3 consecutive days to achieve symptomatic relief. Once complete relief of symptoms has been achieved, treatment should be discontinued. Treatment should not exceed 4 weeks without consulting a doctor. If no improvement in symptoms is observed within 2 weeks of continuous treatment, the patient should contact their doctor. Special populations No dose adjustment is necessary in elderly patients or in patients with renal or hepatic impairment. Paediatric use The use of MAALOX REFLUSSO is not recommended in children and adolescents under 18 years of age due to insufficient data on safety and efficacy. Method of administration MAALOX REFLUSSO 20 mg gastro-resistant tablets should not be chewed or crushed, and should be swallowed whole with liquid before a meal.
Warnings and precautions
Patients should be instructed to contact their doctor if: they have unintentional flatulence, anemia, gastrointestinal bleeding, dysphagia, persistent vomiting or vomiting blood, as the medicine may alleviate symptoms and delay the diagnosis of a serious condition. In these cases, malignancy must be ruled out. Have had previous gastric ulcers or gastrointestinal surgery. Are receiving continuous symptomatic treatment for indigestion or heartburn for 4 weeks or more. Have jaundice, liver impairment, or liver disease. Have any other serious medical condition that affects general well-being. Are over 55 years of age with new or recently changed symptoms. Patients with recurrent symptoms of indigestion or heartburn should consult their doctor at regular intervals. In particular, patients over 55 years of age who take any non-prescription medicines for indigestion or heartburn on a daily basis should inform their pharmacist or doctor. Patients should not take any other proton pump inhibitors or H2 antagonists at the same time. Patients who are to undergo endoscopy or breath tests should consult their doctor before taking this medicine. Patients should be advised that the tablets are not intended to provide immediate relief. Patients may begin to experience an improvement in their symptoms after about one day of treatment with pantoprazole, but may need to take it for 7 days to achieve complete heartburn control. Patients should not take pantoprazole as a preventive medication. Decreased gastric acidity, due to any cause – including proton pump inhibitors – increases the gastric counts of bacteria normally present in the gastrointestinal tract. Treatment with acid-reducing medicines causes a slightly increased risk of gastrointestinal infections such as Salmonella, Campylobacter, or C. difficile. This medicine contains maltitol. Patients with rare hereditary problems of fructose intolerance should not take this medicine. This medicine contains lecithin derived from soya oil. If the patient is allergic to peanuts or soya, he/she should not use this medicinal product.
Interactions
MAALOX REFLUX may cause heartburn. Reduce the absorption of active substances whose bioavailability is dependent on gastric pH (e.g., ketoconazole). Co-administration of atazanavir 300 mg/ritonavir 100 mg with omeprazole (40 mg once daily) or atazanavir 400 mg with lansoprazole (60 mg single dose) in healthy volunteers has been shown to result in a substantial reduction in the bioavailability of atazanavir. The absorption of atazanavir is pH-dependent. Therefore, pantoprazole should not be administered concomitantly with atazanavir (see section 4.3). Pantoprazole is metabolised in the liver via the cytochrome P450 enzyme system. An interaction of pantoprazole with other substances metabolised by this enzyme system cannot be ruled out. However, no clinically significant interactions were observed in specific tests with carbamazepine, caffeine, diazepam, diclofenac, digoxin, ethanol, glibenclamide, metoprolol, naproxen, nifedipine, phenytoin, piroxicam, theophylline, and an oral contraceptive containing levonorgestrel and ethinyl estradiol. Although no interactions were observed in clinical pharmacokinetic studies during concomitant treatment with phenprocoumon or warfarin, a few isolated cases of changes in International Normalized Ratio (INR) have been reported during concomitant treatment in the post-marketing period. Therefore, in patients treated with coumarin anticoagulants (e.g., phenprocoumon or warfarin), it is recommended to monitor the prothrombin time/INR when pantoprazole is initiated, discontinued, or administered intermittently. No interactions have been observed with concomitantly administered antacids.
Price Trend
This product has been on sale since 25/09/2017
In the last 30 days, the product's lowest price was 5,85 €