Free shipping from 69,00 €
Get free shipping with a minimum spend of 69,00 €. Or by purchasing one of the selected products. Discover them now!
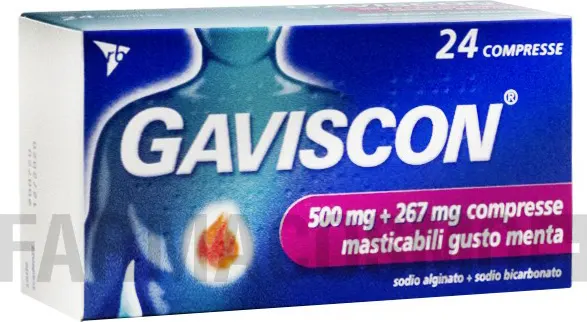
Gaviscon 500 mg + 267 mg - counteracts the occasional heartburn mint aroma 24 chewable tablets
Information on returns and shipments
Payment methods
Medicines authorized by the Ministry of Health

Symptomatic treatment of occasional heartburn.
Indications
Gaviscon 500mg + 267mg mint-flavored chewable tablets, neutralises excess stomach acid and forms a protective barrier that floats on the stomach contents, preventing their reflux.
Mint flavor.
Composition
Active ingredients
Gaviscon 500 mg + 267 mg mint-flavored chewable tablets One tablet contains: Active ingredients: sodium alginate 500 mg; sodium bicarbonate 267 mg. Excipient with known effect: aspartame 7.5 mg.
Excipients
Mint-flavored chewable tablets Mannitol (E421), calcium carbonate, magnesium stearate, copovidone, aspartame (E951), acesulfame potassium, macrogol 20,000, mint flavor.
Directions for use and dosage
Adults and adolescents (12-18 years): 10-20 ml (second-fourth notch of the measuring cup or 2-4 measuring spoons or 1-2 single-dose sachets) after meals and at bedtime.
Method of administration: For oral administration.
Shake the suspension before use.
Warnings
Do not use for prolonged treatment.
In adolescents (12–18 years), use only if clearly needed and under strict medical supervision. Renal impairment In case of renal impairment, the medicine should be used with caution since the exogenous salt supplement provided by antacids can cause potentially serious electrolyte imbalances. Each 500 mg + 267 mg tablet contains 123 mg (5.3 mmol) of sodium.
This should be taken into consideration in cases where a particularly low-salt diet is recommended, for example in some cases of congestive heart failure and renal impairment.
Each 500 mg + 267 mg tablet contains 160 mg (1.6 mmol) of calcium carbonate.
Caution should be exercised when treating patients with hypercalcaemia, nephrocalcinosis, and recurrent calcium-containing kidney stones.
The tablets and oral powder contain aspartame, a source of phenylalanine: it should therefore not be taken by patients with phenylketonuria.
The suspension contains methyl parahydroxybenzoate and propyl parahydroxybenzoate, which may cause allergic reactions.
Delayed reactions (contact dermatitis) generally occur, rarely immediate reactions with urticaria and bronchospasm.
Duration of treatment: If symptoms do not improve after seven days, the clinical picture should be re-evaluated.
Contraindications
Hypersensitivity to the active substances or to any of the excipients such as methyl parahydroxybenzoate (E218) and propyl parahydroxybenzoate (E216) (parabens).
Interactions
It is advisable to leave an interval of at least two hours between taking Gaviscon and that of other drugs, especially tetracyclines, fluoroquinolones, iron salts, thyroid hormones, chloroquine, bisphosphonates and estramustine.
Undesirable effects
The following are the undesirable effects of Gaviscon, organised according to the MedDRA system organ classification.
They are divided by frequency (very common (≥ 1/10), common (≥ 1/100 to ≤ 1/10), uncommon (≥ 1/1,000 to ≤ 1/100), rare (≥ 1/10,000 to 1/1,000), very rare (≤ 1/10,000)).
| System Organ Class | Frequency | Adverse Reaction |
| Immune system disorders | Very rare | Anaphylactic or anaphylactoid reactions. Hypersensitivity reactions (such as urticaria). |
| Respiratory, thoracic and mediastinal disorders | Very rare | respiratory symptoms such as bronchospasm |
| Gastrointestinal disorders | Very rare | flatulence, nausea |
| General disorders and administration site conditions | Very rare | oedema |
Overdose
Experience with overdose is very limited.
The only possible consequence of overdose is abdominal distension: in this case, resort to symptomatic treatment and adopt general supportive measures.
Pregnancy and breastfeeding
Use only if clearly needed and under strict medical supervision.
Storage
Do not store above 25°C. Store in the original packaging. Do not refrigerate. Mint-flavored chewable tablets: do not store above 30°C. Store in the original packaging.
Format
24 Chewable Tablets
Price Trend
This product has been on sale since 09/11/2017
In the last 30 days, the product's lowest price was 8,72 €